Winnonlin training course
- #Winnonlin training course how to#
- #Winnonlin training course manual#
- #Winnonlin training course software#
- #Winnonlin training course license#
- #Winnonlin training course series#
#Winnonlin training course software#
Licenses can be obtained from the software vendor, Certara.
#Winnonlin training course license#
Note: Students must provide their own license for Phoenix WinNonlin. Register now and learn to use Phoenix WinNonlin. Register now and learn the same information using a resource that will be available to you 24 hours a day, 7 days a week, with lifetime access.
#Winnonlin training course how to#
All lectures are screencasts showing how to use Phoenix tools. We offer training in pharmacology and toxicology to both undergraduate and.

The entire course is a hands-on practical course. This unique course harbors the state of the art information on toxicology. The objective of the course is to increase the productivity and efficiency by introducing participants to numerous tools and techniques available in Phoenix WinNonlin. This course is suitable for beginning scientists, experienced pharmacokineticists, toxicokineticists, clinical pharmacologists, and anyone who uses handles pharmacokinetic or pharmacodynamic data. This course provides a comprehensive overview of the basic functions within Phoenix WinNonlin to perform noncompartmental or compartmental analysis.

This course will demonstrate how to use Phoenix WinNonlin to perform pharmacokinetic analysis, and how the Phoenix platform can create efficiency and reproducibility in analyses. While all of the same functionality exists, many users find the Phoenix platform difficult to navigate.
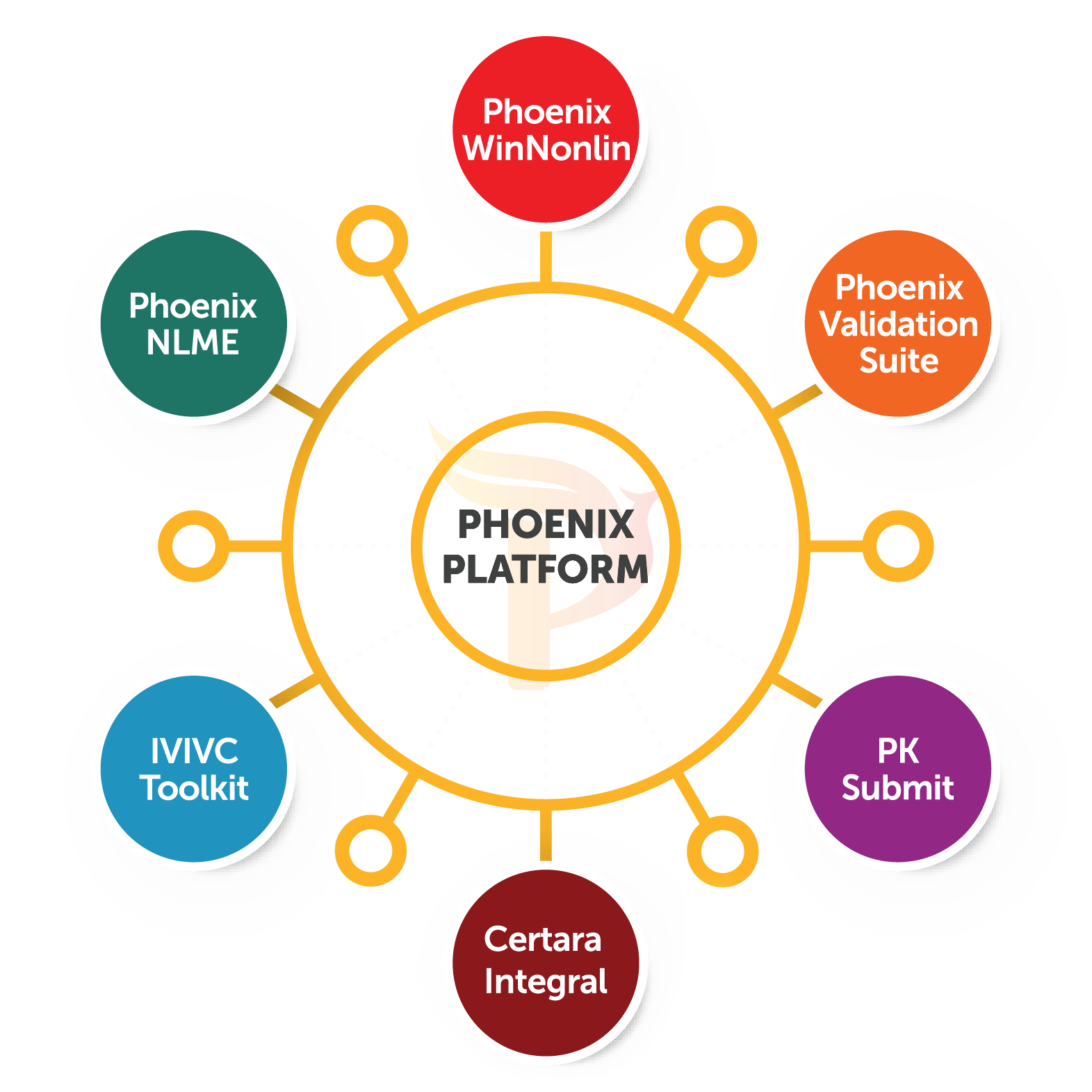
However, the new Phoenix platform is a drastic departure from the previous versions of WinNonlin. Here, we provide a step-wise illustration of how to perform a calculation of microsomal clearance from UNIFI data acquired on Xevo G2-S QTof using WinNonlin.As the industry standard for pharmacokinetic analysis, Phoenix WinNonlin is a key tool for the pharmacokinetic scientists. All of these features help to minimize errors and enable the more confident analysis of large data sets.
#Winnonlin training course manual#
The exported data can be brought into third-party packages without manual copying or transcription and the table layouts preferences can be saved within UNIFI. Flexibility in defining and customizing columns to be exported (i.e.: filter data performed in human species at a specific dose or time points). Entire processed datasets or filtered subsets data (such as separate tables for QCs, Unknowns, etc.).Summary and custom calculations (support for built in UNIFI calculations such as max, min, average values, etc.).Custom columns (user customizable values).Specific columns and column order (such as response, response ratio, time, species, treatments, subject ID, etc.).The export functionality can be tailored to include specific information, such as: Tables in UNIFI can be configured and saved to export exactly what the user needs.

xls support and limited support for open standard spectral. Direct export of tabular data (comprehensive Excel.The blood-tissue exchange of each organ is characterized by its volume, perfusion, metabolism, capillary permeability and blood/tissue partition coefficient. Electronic and customizable data transfer/integration between instrumentation software and third-party software platforms (WinNonlin, Spotfire, etc.) is the preferred approach and is essential for handling and processing large amounts of data with both accuracy and speed.Įnhanced export using UNIFI Software provides the following features: Physiologically based pharmacokinetics (PBPK) uses a realistic organ model to describe drug kinetics.
#Winnonlin training course series#
Calculation of PK parameters are commonly performed to establish the viability of a drug candidate or series and help prioritization efforts. It is critical for LC-MS software to support easy transfer of results to a variety of third-party software packages in order to extract and provide key information to support drug discovery and development.Īn example of this is to use LC-MS data to calculate pharmacokinetics parameters using industry-standard software packages such as Phoenix WinNonlin (Certara, St. How we convert this data to drive decisions is often the bottleneck as many different software tools are used in a typical DPMK lab. Generating LC-MS data is an integral part of drug metabolism pharmacokinetic (DMPK) departments.
